Enable Regulated Bioinformatics
From preclinical discovery to clinical trials and commercial manufacturing, Seqera enables organizations to run validated bioinformatics workflows with GxP-grade quality.
Request a DemoSeqera Platform now serves as a validated orchestration layer for regulated bioinformatics pipeline execution that is a part of the manufacturing process, supporting ongoing clinical trials. The deployment is designed to scale seamlessly from clinical development through commercial manufacturing, positioning it for use in GMP operations post-BLA approval.
Seqera Platform now serves as a validated orchestration layer for regulated bioinformatics pipeline execution that is a part of the manufacturing process, supporting ongoing clinical trials. The deployment is designed to scale seamlessly from clinical development through commercial manufacturing, positioning it for use in GMP operations post-BLA approval.
— Albert Park, Head of Digital for Research and Tech Dev

From Discovery to Regulatory Submissions
Seqera already partners with leading pharma companies across the drug development lifecycle, powering large-scale deployments that demand traceability, control, and reproducibility. As organizations advance critical programs from discovery through regulatory submissions, Seqera's GxP-offering enables validated bioinformatics workflows that deliver secure, traceable, and scalable analysis aligned with FDA and EMA expectations.
Data Access & Management
Unified access and fine-grained control over research and clinical data, making it faster and easier to launch analyses.
Scalable Data Analysis
Secure, scalable execution, from exploratory, AI-driven workflows to fully validated production pipelines.
Validated Workflows
Standardize and automate complex data processes, improving reproducibility and enabling reuse across studies and teams.
Security & Compliance
Validate faster with built-in compliance features, documentation, and validation services to expedite your journey to GxP.
Why Seqera for Regulatory Bioinformatics
Built on high-quality enterprise software, our approach combines proven enterprise architecture with deep bioinformatics expertise, ensuring your teams can maintain velocity while meeting the most stringent compliance requirements. With native support for audit logging, immutable versioning, and role-based access control, Seqera provides the foundation for validated workflows that scale with your science.
GxP-Ready Design
Purpose-built to support current Good Laboratory Practices (cGLP), current Good Clinical Practices (cGCP), and current Good Manufacturing Practices (cGMP) environments with GAMP5-aligned documentation, comprehensive controls, and validation-ready architecture.

Your Infrastructure
Deploy workflows directly in your environment without moving data. Self-hosted architecture integrates seamlessly with existing IAM, storage, and networking while capturing complete data and compute provenance.
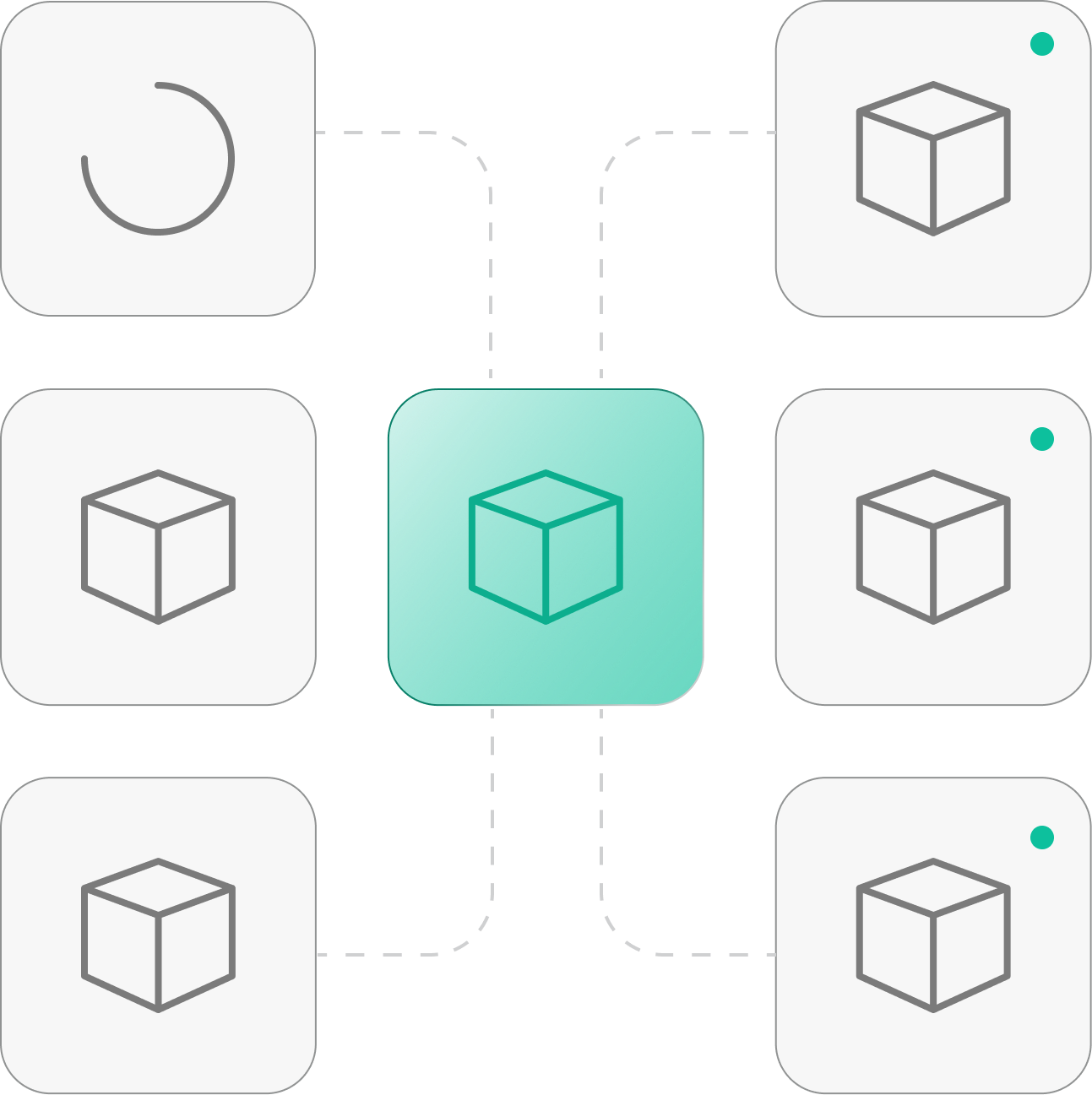
Full Traceability & Reproducibility
Complete workflow provenance captures inputs, steps, environments, and outputs. Every analysis is reproducible and audit-ready with full run history from research through clinical and manufacturing environments.
Audit-Ready Operations
Human-readable audit logs with comprehensive, 21 CFR § 11- and Annex 11-aligned, and inspection-ready change tracking.
Enterprise-Grade Quality
SOC2 Type II attestation report covers the product and supporting operations with multi-layered security across product, infrastructure, and organizational levels.

Accelerated Validation
Pre-built validation templates, test protocols, generic test evidence, and risk-based planning aligned with GAMP5 2nd Edition; with flexibility to align with your preferred validation frameworks and partners.

How Seqera Powers GxP-Enabled Innovation
Seqera unifies data, workflows, compute, and compliance into a single GxP-ready platform, enabling validated bioinformatics workflows without compromising control or traceability. From preclinical studies to manufacturing quality control, our platform captures complete workflow provenance while integrating directly with your existing infrastructure. Teams gain confidence to innovate at scale, knowing every analysis is reproducible, auditable, and inspection-ready.
Scalable Bioinformatics with Regulatory-Grade Quality
Streamline the path from research to regulatory submissions with a platform built with compliance in mind. Seqera delivers the traceability, control, and validation support required for regulated environments, from preclinical studies to commercial manufacturing.
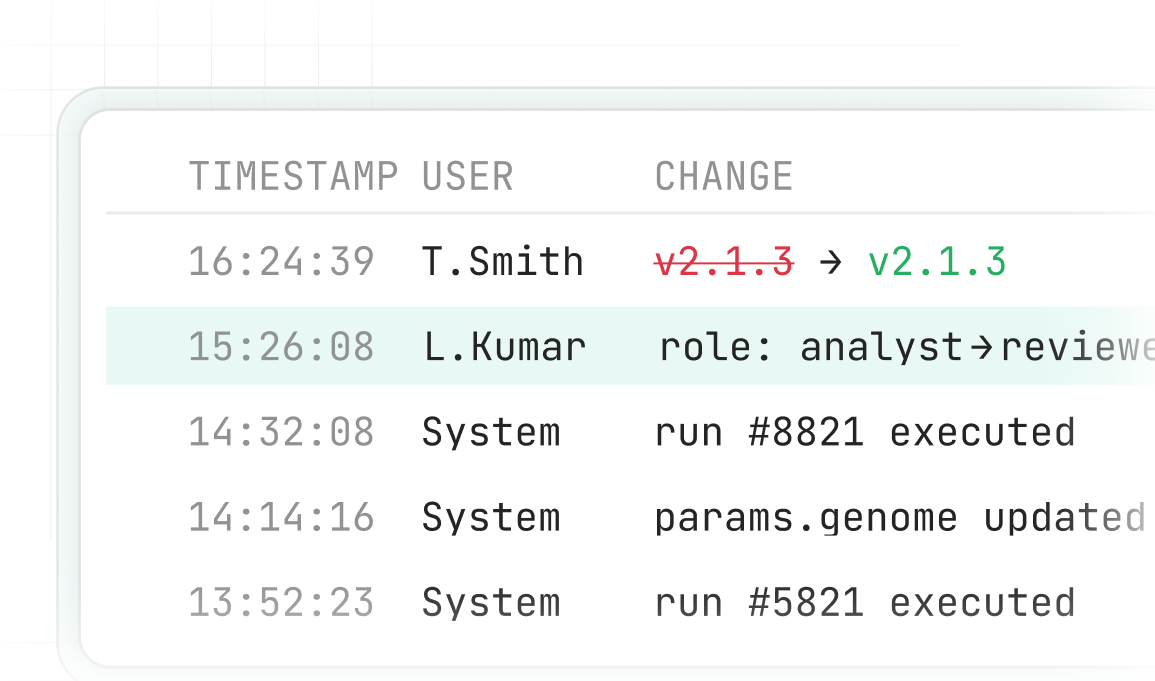
Comprehensive Audit Logs
21 CFR § 11- and Annex 11-aligned audit logging with pre/post-change capture, ensuring complete traceability for inspections and regulatory expectations.
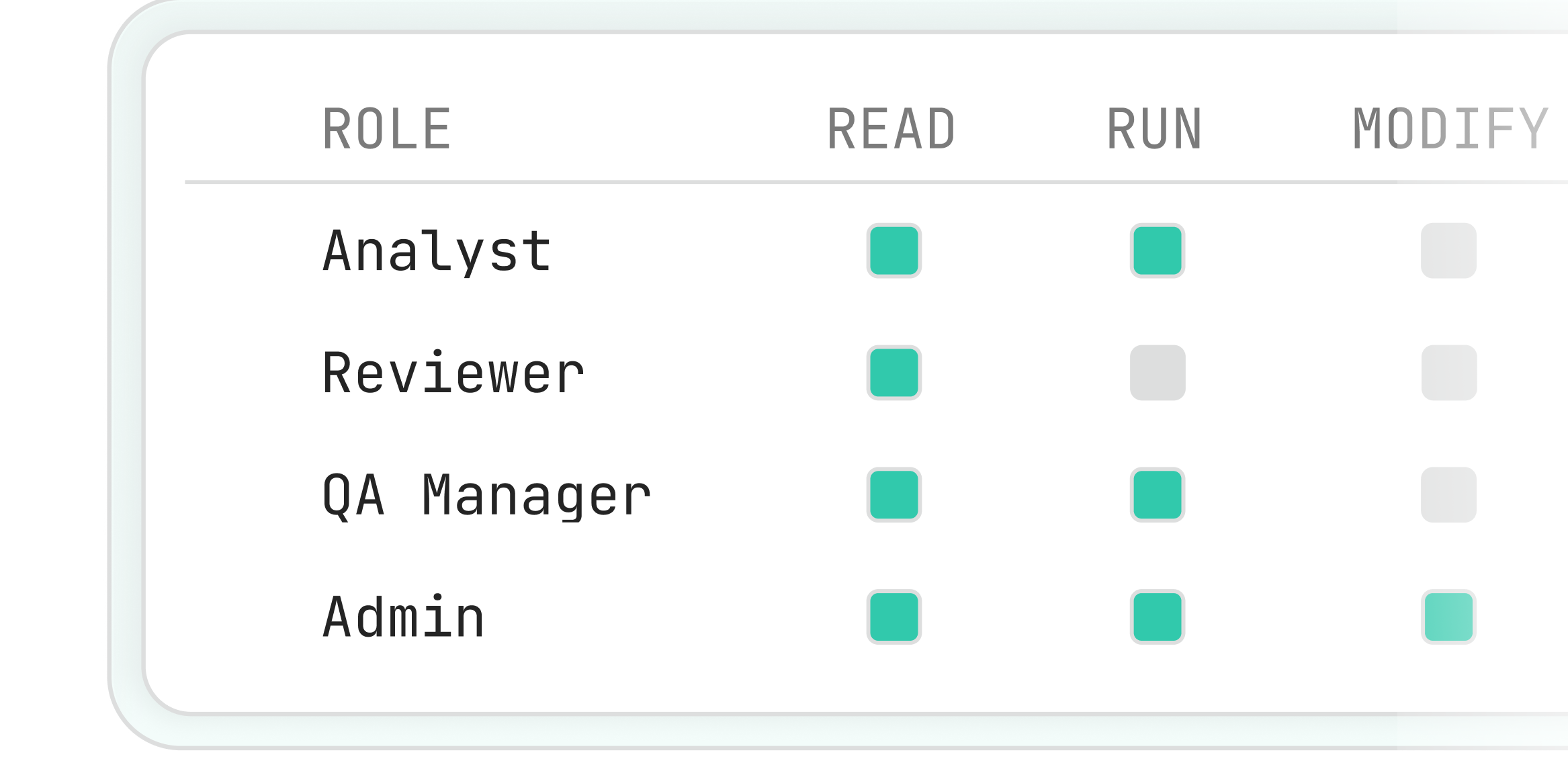
Role-Based Access Control
Fine-grained access controls ensure only authorized users can modify or execute workflows, supporting segregation of duties across cGLP, cGCP, and cGMP environments.

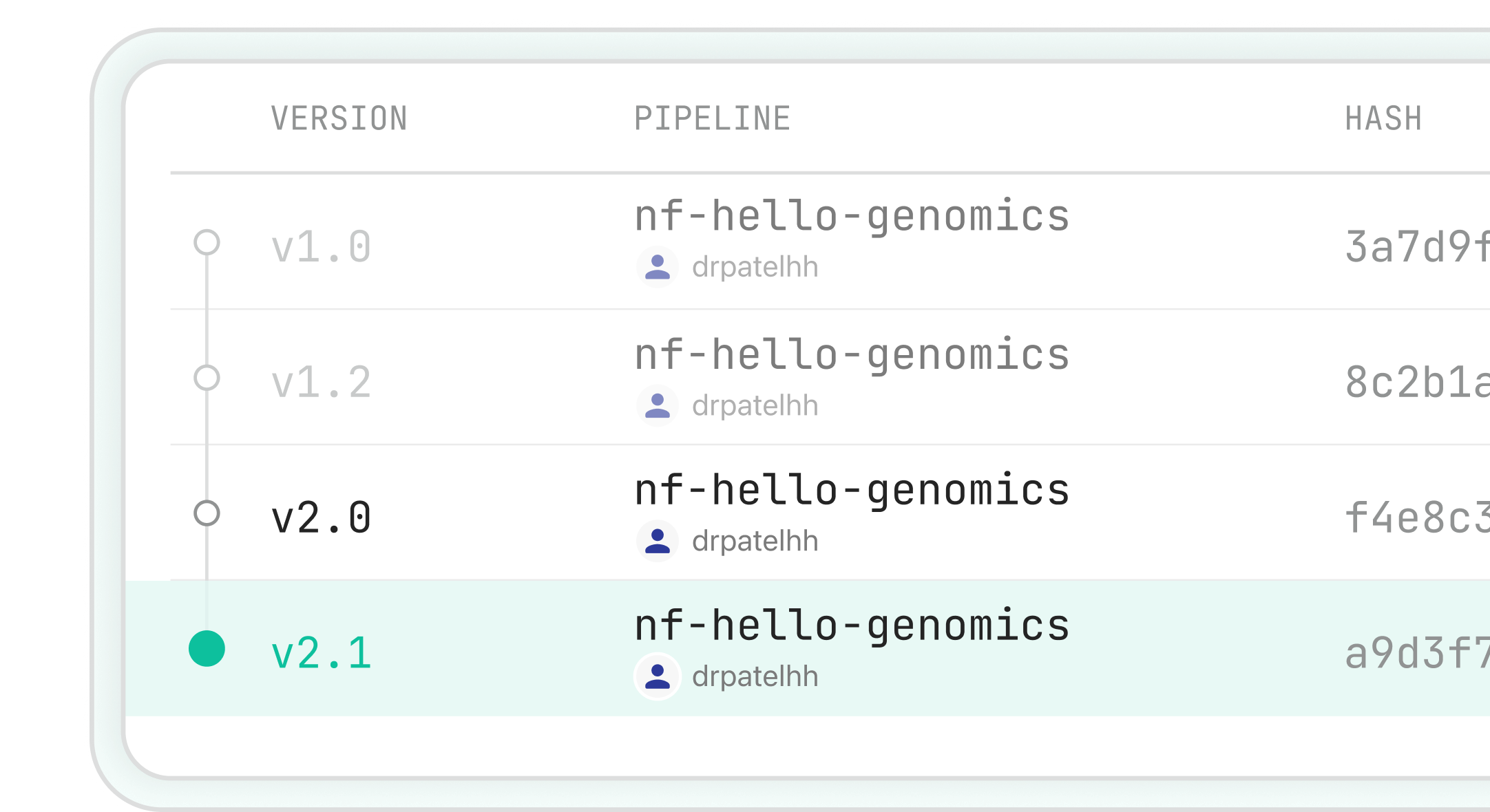
Immutable Workflow Versioning
Git-based version control with immutable pipeline versions ensures every workflow is traceable, reproducible, and audit-ready.
Complete Workflow Provenance
End-to-end tracking captures inputs, processing steps, container environments, and outputs, delivering full data lineage for regulatory submissions and quality control.
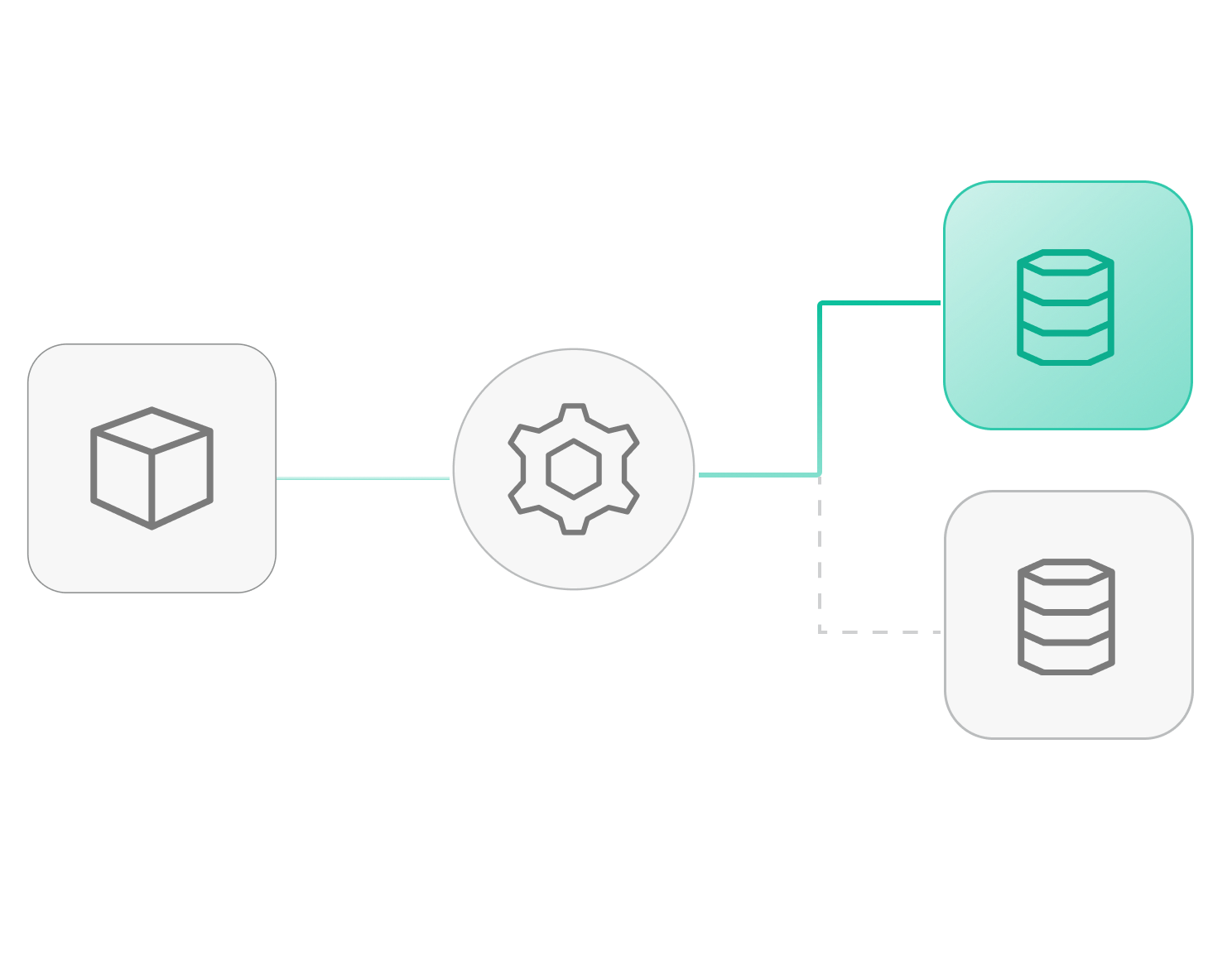
Self-Hosted Architecture
Deploy on your infrastructure without moving data. Your deployment stays under your governance while integrating with existing IAM, storage, networking, and monitoring systems to maintain security and control.
GAMP5-Aligned Validation Support
GAMP5-aligned requirements specs, test cases, and traceability matrix templates accelerate validation while maintaining flexibility with your preferred frameworks and validation partners. Generic test evidence accelerates the validation of your particular configuration and pipelines.

Trusted by 14 of 20 Top Pharma Companies
The world's leading pharma companies trust Seqera to power their most critical workflows across discovery, clinical trials, and manufacturing. Our platform enables teams to maintain scientific agility while meeting stringent regulatory requirements, supporting everything from breakthrough therapeutics to large-scale production environments.
Read more